Renewable energies are energies of peace, freedom and democracy
- Details
EU must increase target for energy efficiency / Money for energy imports to a blocked special account
“Renewable energies are energies of peace, freedom and democracy. We see that very clearly in the current crisis surrounding the terrible war that Putin's army is waging in Ukraine. The European Union imports gas, coal and oil worth 99 billion euros every year.[1] That is almost double the Russian military budget. Consequently, we are financing the war with our money for energy and should therefore not be afraid of blocking Russian supplies of oil, gas and coal, but should be proactive in this regard. The least that must happen is that we send the money for these deliveries to a blocked special account and only release them when the war is over. If in doubt, we have to stop the exports. I find it unbearable that Gazprom Bank is exempt from sanctions against Russian banks. We need to become more independent as soon as possible. This requires short-term compromises, e.g. more use of coal from inside the European Union or countries like South Africa. In the medium and long term, the expansion of renewable energies and more energy efficiency is the only right solution”, emphasized the environmental spokesperson of the EPP group in the European Parliament, Peter Liese.
He emphatically welcomed the proposal by his EPP group colleague Markus Pieper to increase the renewable energy target for the EU from 40 to 45 percent and called for a comparable step in the area of energy efficiency. The ENVI Committee of the European Parliament is discussing its opinion on energy efficiency this Thursday.
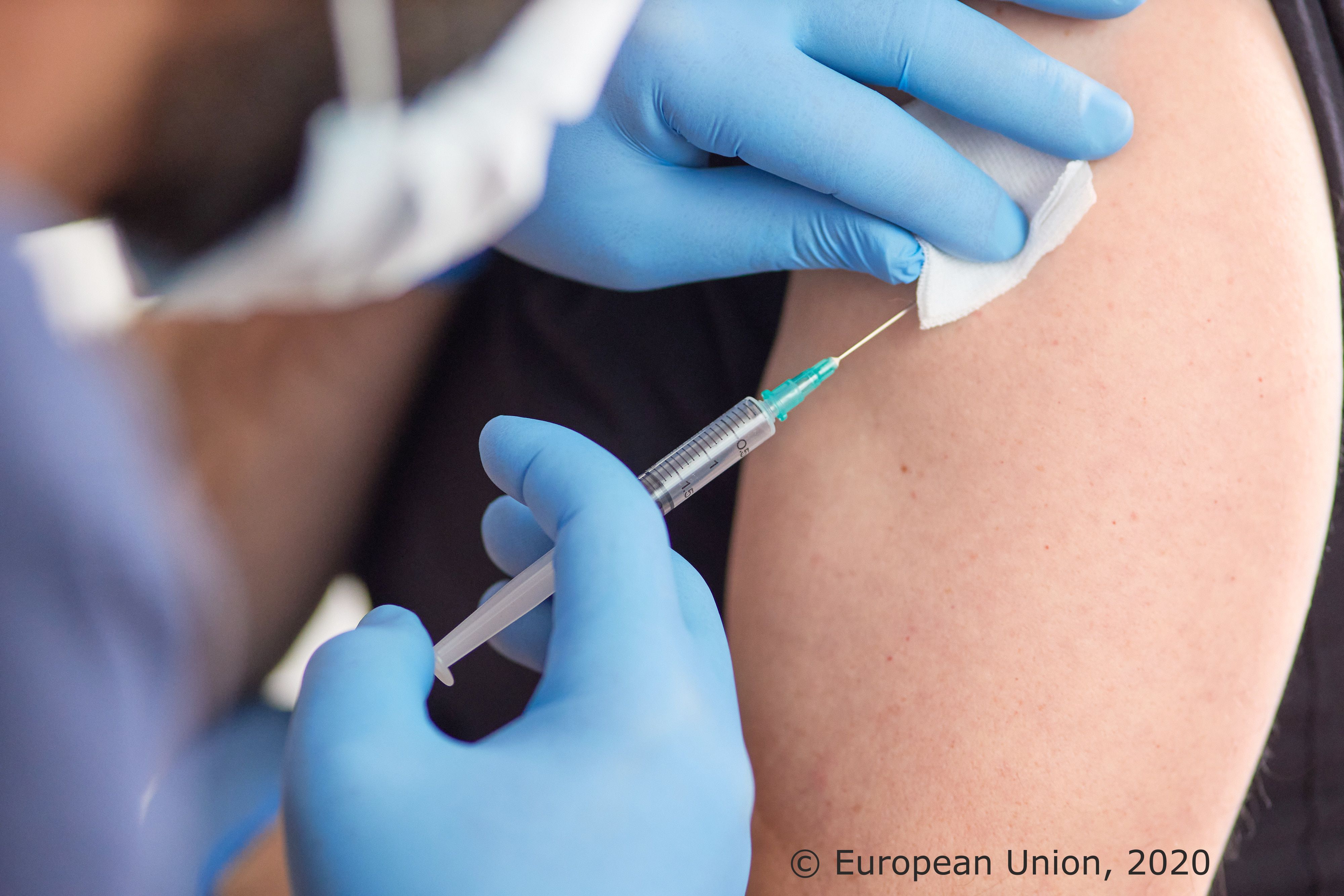
Novavax can help to convince vaccine hesitators and can make a big contribution to vaccination programs in low- and middle- income countries
- Details
European authorisation System has proven helpful for the new company / first doses are leaving the distribution centre in the Netherlands in these minutes
“The new vaccine of Novavax can help to convince people that have not been ready to get vaccinated so far because it is based on a more traditional procedure and helps to speed up the vaccination campaign in low- and middle- income countries.” This concluded the health spokesperson of the biggest group in the European Parliament (EPP Christian Democrats), Peter Liese after a videoconference with the company.
Novavax announced the start of shipments of its vaccine that has already been approved in Europe at the end of last year. “I strongly defend the European authorisation system and all vaccines that get conditional marketing authorisation are safe. The benefits by far outweigh the risks. However, some people have unfortunately not yet been vaccinated because they do not trust the m-RNA and/or the vector technology. They now have an alternative with the protein-based vaccine of Novavax. On top of that, Novavax has much less demanding criteria for storage and the cold chain. That is why it is a good alternative for low- and middle-income countries”, announced Liese.
are paying for the war with our energy bill
- Details
Do EVERYTHING to become more independent from Russia / Compromises on environmental protection necessary in the short term / Keep climate protection targets at all costs, because in the medium and long term climate protection and independence require the same measures / Do not ignore IPCC
"With our energy bill we are financing Putin's war in Ukraine. That is why we must immediately use ALL! alternatives," said the environmental spokesman of the largest group in the European Parliament (EPP, Christian Democrats) Dr Peter Liese on the occasion of the Russian war of aggression in Ukraine. "The world has changed and therefore every policy area must also change. This also applies to environmental policy. At the same time, we must not lose sight of the fact that climate protection requires to a large extent the same measures as the goal of becoming less dependent from Russia. The alarming statements of the IPCC must not be ignored," said Liese.
Liese called for an import ban on coal from Russia in the short term: "Coal is the dirtiest form of energy, and the fact that we are making ourselves dependent on Russia here when there are numerous alternatives, such as South Africa, Australia and Colombia, has been incomprehensible to me for a long time." He referred to calculations according to which the European Union imports energy, i.e. oil, gas and coal, worth 67 billion US dollars annually from Russia. Russia's military budget, on the other hand, is "only" 61 billion US dollars. "Every effort is justified to reduce this high sum and this is where we have to make compromises in the short term, even as environmental politicians. The rejection of fracking must be put to the test, as must the phase-out of nuclear energy in Germany, whereby it must be carefully examined whether the continued operation of the power plants is practically necessary at all. In parts of the European Union, of course, there is still a dependence on Russia, e.g. through spare parts for older Russian-designed power plants."
European Parliament votes on the final report of Special Committee on Beating Cancer
- Details
European cooperation in the fight against cancer indispensable / Remove hurdles for researchers / Cancer risk from alcohol recognised
The European Parliament yesterday voted on the final report of the Special Committee on Beating Cancer with huge majority (652-15-27)
"We want to make life easier for all those fighting cancer, especially patients and researchers," said MEP and health policy spokesperson of the largest group in the European Parliament (EPP-Christian Democrats) Peter Liese. "Cross-border research is essential to fight cancer. This applies to all types of cancers, but particularly the rare ones as well as paediatric cancer." Liese explained that it is only through European cooperation that sufficient patients can be gathered quickly enough, for example for clinical trials, in order to introduce innovation in the market in a timely manner. Researchers, however, suffer from excessive bureaucracy and different regulations in the Member States. "Change is needed urgently" says Liese. "We need a uniform implementation of the General Data Protection Regulation and a structure in the European Commission to which researchers can turn when they encounter obstacles, in order to then jointly remove them. Both points are included in the text of the Special Committee adopted by plenary. We want to facilitate cross-border research, rolling out the red carpet for researchers, rather than putting obstacles in their way" says Liese. In addition, MEPs call for tailor-made incentives to promote the development of medical drugs treating paediatric cancer as well as incentives to facilitate access to cross-border clinical trials," says Liese, who himself worked as a doctor in a paediatric clinic.
Read more: European Parliament votes on the final report of Special Committee on Beating Cancer