We must curb rise in electricity prices - Main problem, however, remains dependence on gas
- Details
Lead MEP proposes stronger regulation of emissions trading / Market reacts immediately
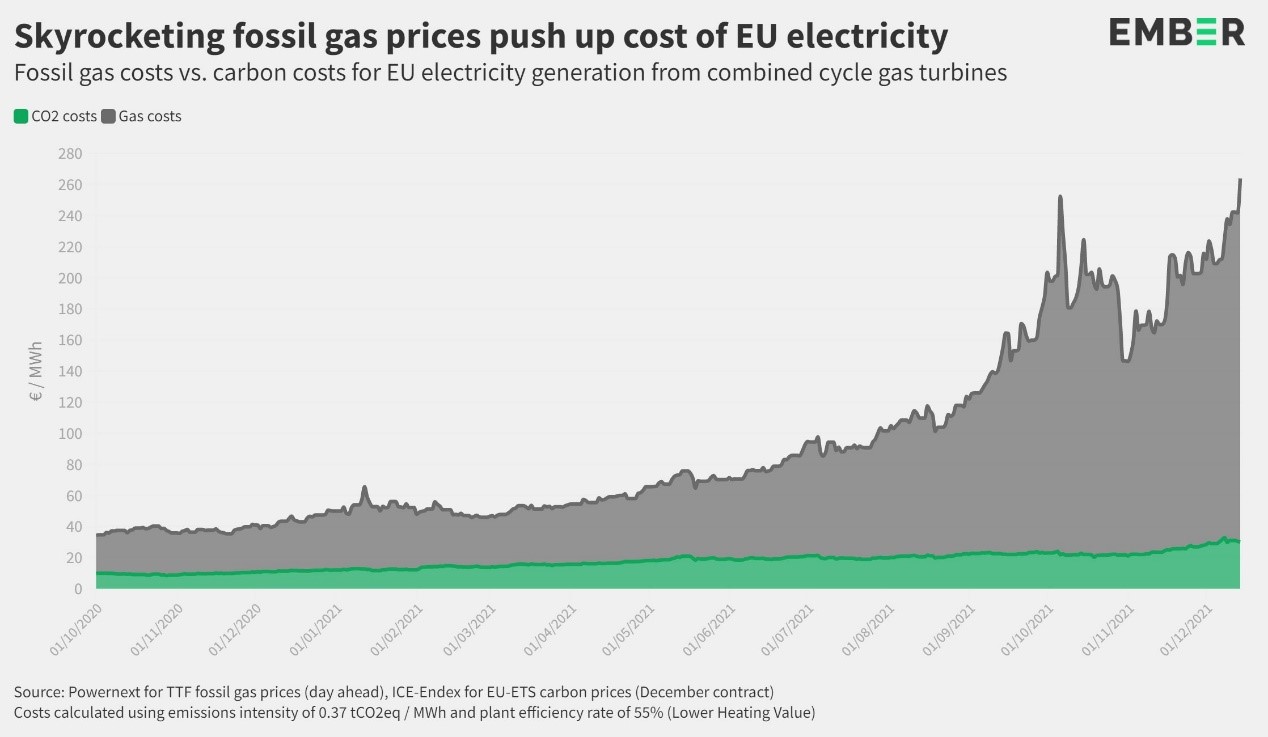
The EPP MEP Dr Peter Liese, as lead rapporteur, proposed amendments for the stronger regulation of emissions trading. As a result, the market reacted immediately with the ETS price falling. According to experts, this will curb the rise of electricity prices across Europe. "High energy prices are a major problem for many families, especially low-income ones, as well as businesses, particularly SMEs. Hence, immediate action is needed. For me it is very clear: the biggest problem is our dependence on fossil fuels, especially Russian gas. Following many discussions with experts, it is clear that Russia is deliberately driving up the price of gas and this is not only a problem for gas consumers, but also the main reason for the rise in electricity prices. Therefore, it is imperative to accelerate the transition to renewable energy and ensure energy efficiency. The Green Deal is the solution, not the problem. Nevertheless, immediate action is required, which is why I have put forward a 4-point plan that includes the following steps:
1. Expanding renewable energies and energy efficiency
2. Lowering taxes and levies, especially on electricity
3. Curbing the ambitions of the EU ETS
4. Introducing regulation for the market of emissions trading certificates."
Maritime ETS: high time for carbon pricing
- Details
IMO failed / Targeted use of the revenues necessary
"It is high time for carbon pricing in the maritime sector,” said Peter Liese, rapporteur for the EU ETS revision in advance of the presentation of his Draft report in the ENVI committee on Thursday. “The European parliament has continuously asked for including the maritime sector in the ETS, and it is good that the Commission included in its proposal. I would prefer a global solution, but we clearly have to state that IMO failed to address the problem. This is also shared by companies in this sector.”
Dr. Reinhard Lüken, Managing Director of the Shipbuilding and Marine Engineering Association (VSM) agreed: “IMO member states have so far missed every opportunity to collectively send credible positive signals for determined climate action. Therefore, although we prefer global provisions as a global industry, we are now convinced that the EU must lead the way for the maritime industry if we are to achieve results in time. If the EU succeeds in combining ambitious climate protection with economic success, the European model will quickly find imitators and thus promote comprehensive global progress.”
European Parliament to vote on the final report of Special Committee on Beating Cancer
- Details
European cooperation in the fight against cancer is indispensable / Making life easier for all those fighting cancer / Red carpet for cross-border research / Concrete measures must follow
Tomorrow the European Parliament will vote on the final report of the Special Committee on Beating Cancer. Since 2020, the fight against cancer has been a priority for the European Union. The Christian Democratic EPP group had convinced Commission President Ursula von der Leyen and successfully demanded a mandate for this Special Committee. "We want to make life easier for all those who are fighting cancer, especially patients and researchers," says MEP and health policy spokesperson of the largest group in the European Parliament (EPP-Christian Democrats) Dr med. Peter Liese. "Cross-border research is essential to fight cancer. This is true for all cancer types, but especially for the rare ones as well as those impacting children." According to Prof Angelika Eggert, Director of the Clinic for Paediatrics with a focus on oncology and haematology at the Charité in Berlin it is only through European cooperation that sufficient patients, for example for clinical trials, can be gathered quickly enough to introduce innovation in the market in a timely manner. However, researchers suffer from excessive bureaucracy and different regulations across the member states. "Change is needed urgently" says Liese. "We need a uniform implementation of the General Data Protection Regulation and a structure in the European Commission to which researchers can turn when they encounter obstacles, in order to then remove them together”. Both points are included in the text of the Special Committee. We want to facilitate cross-border research, rolling out the red carpet for researchers, rather than putting obstacles in their way" says Liese. In addition, MEPs call for tailor-made incentives to promote the development of medical drugs treating paediatric cancer as well as incentives to facilitate access to cross-border clinical trials," says Liese, who himself worked as a doctor in a paediatric clinic.
Read more: European Parliament to vote on the final report of Special Committee on Beating Cancer
Dependence on fossil fuels and Russian (manipulative) behaviour main cause of high-energy prices
- Details
Green deal is the solution not the cause / However, targeted changes in ETS useful / 4 points plan against high energy prices
“The main cause of the high-energy prices are our dependence on fossil fuels and the behaviour of Russia.” This was clearly expressed by two key MEPs in a video conference for journalists on Wednesday. Peter Liese and Andrius Kubilius explained that the current increase in the energy price including electricity prices is driven by scarcity of gas, and mainly caused because of Russia’s dominating position on the market and EU’s high dependence on Russian gas. Andrius Kubilius presented clear evidence that activities of Russia in European gas markets result in an unprecedented surge of natural gas prices in the EU.
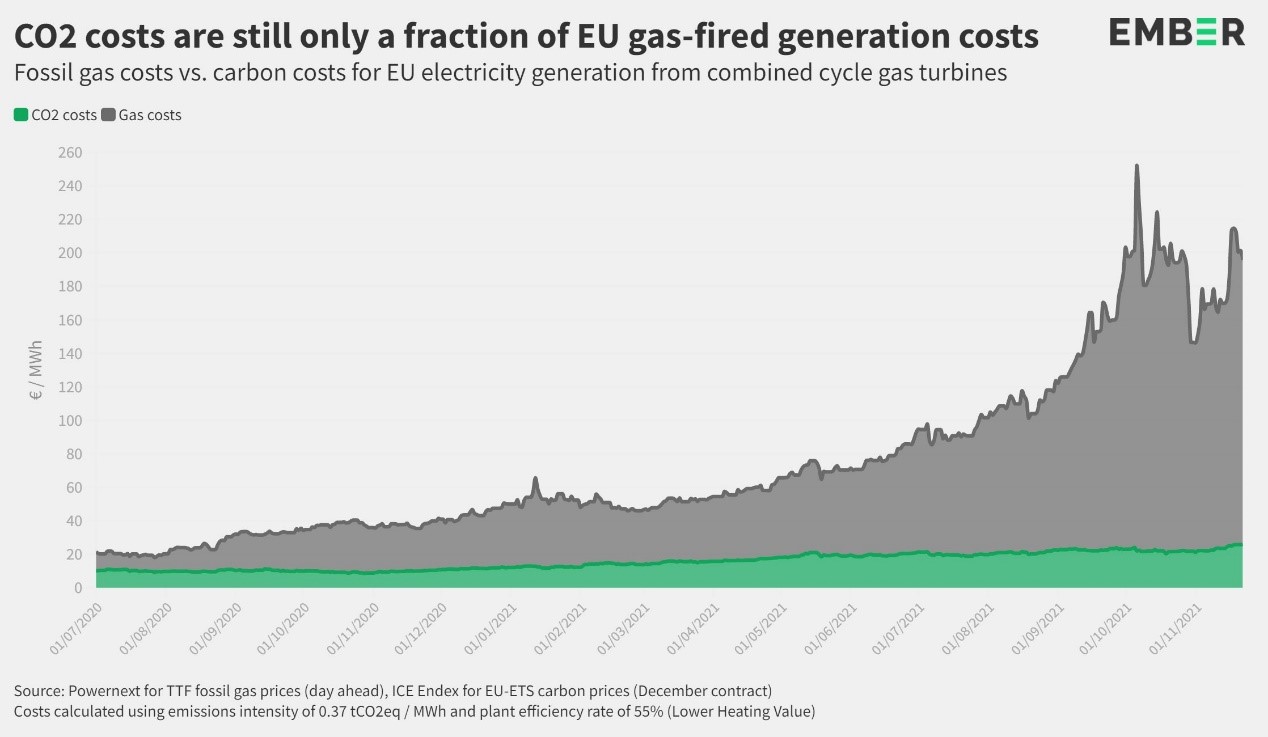
Mr Kubilius says that the current increase in the energy price, including electricity prices, is driven by the artificial crisis in European gas market. The EPP MEP explained that evidence, collected by International Energy Agency, is showing very clearly that Gazprom in the last quarter of 2021 cut the supply of gas to European markets by 24% if to compare with data of 2020. That resulted in a huge increase of gas prices on European markets. According to Andrius Kubilius, the goal of Gazprom is to fight the implementation of the Green Deal, because it would diminish the consumption of gas in Europe by 60-70% in 2035. For the Kremlin that is a very dangerous perspective and they started the war against Green Deal. The increase of prices is not influenced by the implementation of the Green Deal, but by Gazprom fighting against the Green Deal1.